Glowing in the dark bright green sphere and blue egg
- Details
- Created: Sunday, 25 July 2021 09:58
The ornamental egg and sphere shown in figure 1 were bought from China as 'Glowing in the dark egg and sphere'. Without knowing what they really are, they make an interesting case to study, especially for luminescence and phosphorescence. Their color in daylight is as shown in figure 2, i.e. white. The sphere, is never totally white, there is a reminiscent dull light green color. In figure 1, exposed to the LWUV and SWUV, they become blue and bright green (luminescence, often called fluorescence) and remain like that for a very long time, needless to say these colors fade with the time (phosphorescence). The figure 3 shows the decay of the phosphorescence up to 8 minutes but it lasts for more than 12 hours, obviously weak but still visible to the human eye. It has not been experimented to put it in a closed and opaque box and then to observe it every day to check if the phosphorescence is still there.
 Figure 1. The glowing in the dark 90 g egg and 65 g sphere exibiting
Figure 1. The glowing in the dark 90 g egg and 65 g sphere exibitingblue and bright green colors while exposed (luminescence) and after
exposure (phosphorescence) to LWUV.
 Figure 2. The glowing in the dark 90 g egg and 65 g sphere exibiting
Figure 2. The glowing in the dark 90 g egg and 65 g sphere exibitingtheir color under daylight. Note the sphere is still slightly greenish,
there is always a reminiscent green phosphorescence.
The luminescence color is evently spread on the egg and the sphere but some unevenness can be observed, both show darker areas as shown in figure 1. In daylight, these areas are whiter than the surrounding ones. Either the material is not homogeneous or a coating is applied to the surface with some irregularities. To verify this hypothesis, the sphere is ground to get a flat surface and to have a window on what is inside. The ground sphere is shown in figure 4, lighted by LWUV and daylight equivalent lamp. The ground surface id dark and almost inert to the LWUV although the surronding glows. This confirms these objects are coated to produce their glow! In daylight, the material is whiter than the surface of the sphere. The texture looks like a marble with quite large grains that can be observed at the top of the ground surface. The marble theory has to be verified with spectroscopy.
| Shape | egg, sphere (ball) |
| Size | egg: 47 x 36 mm, sphere: Ø 35 mm |
| Color | white |
| Diaphaneity | opaque |
| Lustre | dull |
| Weight | egg: 90 g, sphere: 65 g |
| SG | the egg and the sphere are two heavy for the hydrostatic scale |
| RI | - |
| DR | - |
| Pleochroism | - |
| Polariscope / Conoscope | - |
| SWUV | egg: blue, sphere: bright green |
| LWUV | egg: blue, sphere: bright green |
| Magnetic susceptibility N52 | - |
| Chelsea filter | - |
Table 1. Observational and measured properties
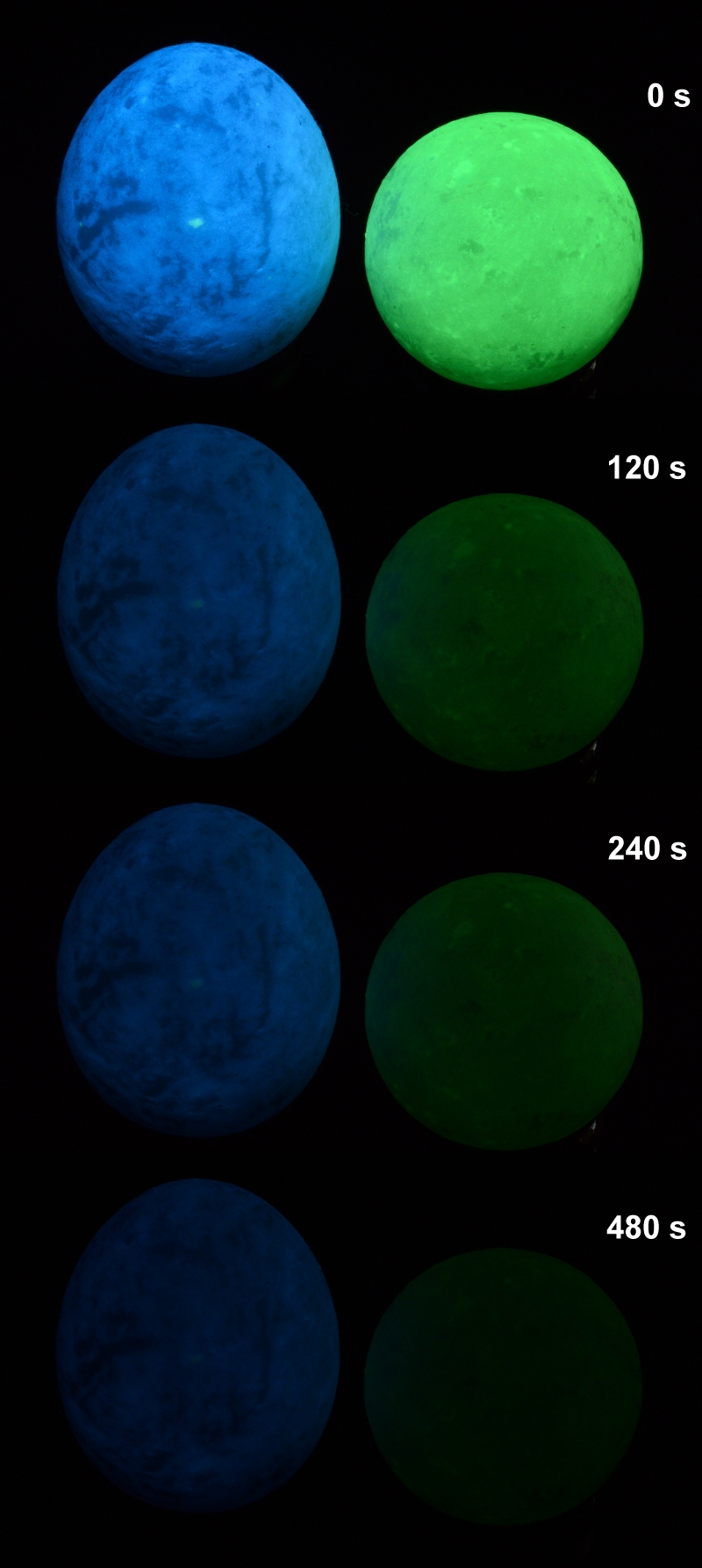
Figure 3. Phosphorescence decay after LWUV exposure photographed over 8 minutes, a picture was taken every two minutes..
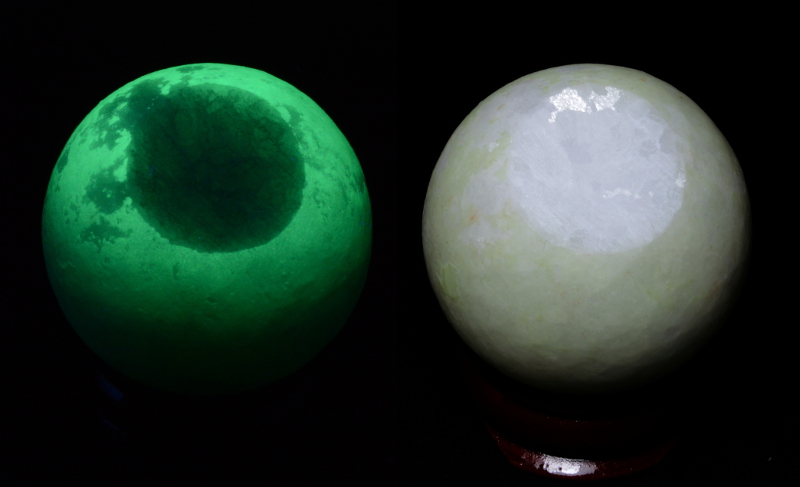
Figure 4. The ground sphere, left, exposed to LWUV and right, lighted with a daylight equivalent lamp. The ground surface does not
luminesce, the suspected coating is now confirmed. It shows a material composed of large white grains as it can be found in some marbles.
Raman spectroscopy:
Several Raman spectra are collected from the flat surface, all yield the same result as shown in figure 5. There has been no attempt at collecting Raman spectra of the luminescent coating. The grains in the white material are dolomite as indicated by their Raman spectra, so the sphere is made of a dolomitic marble. Even if not verified on the egg, it is very likely the same.
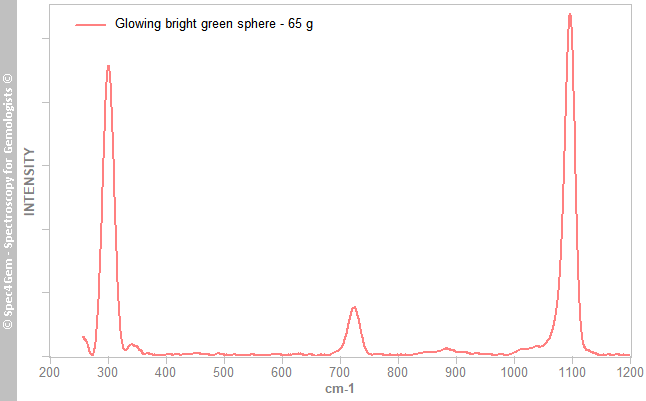 Figure 5. Raman spectra collected from several spot on the ground flat surface of the sphere. Even if the calcite and dolomite have similar spectra, these ones are undoubtedly these of dolomite.
Figure 5. Raman spectra collected from several spot on the ground flat surface of the sphere. Even if the calcite and dolomite have similar spectra, these ones are undoubtedly these of dolomite.Photoluminescence spectroscopy:
The photoluminescence spectra of the sphere and the egg are collected with the common excitations : 254, 310, 370 and 405 nm. The green luminescence spectra of the sphere are shown in figure 6, they are normalized to the same intensity and all are strictly identical with a peak at 522 ± 2 nm. Similarly, the blue luminescence spectra of the egg, as shown in figure 7, yield a peak at 472 ± 2 nm. Note, the green luminescence peak (FWHM ~80 nm) is broader than this of the blue luminescence (FWHM ~60 nm) and is more asymetric with a discreet hump arround 555 nm.
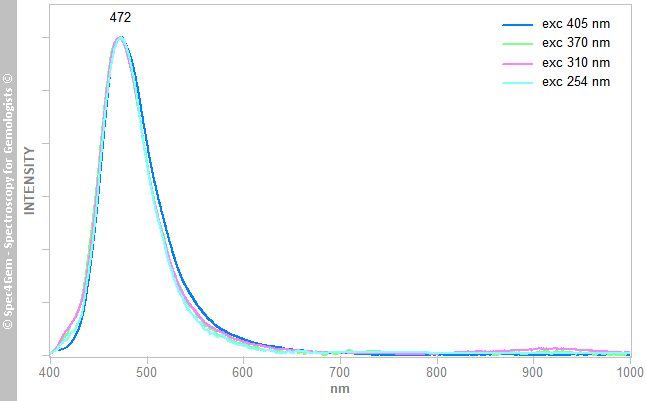 Figure 6. Photoluminescence spectra of the egg showing a strong blue luminescence with a maximum at 472 nm. This luminescence is attributed to the coating suspected to contain SrAl4O7:Eu2+ (Sr4Al14O25:Eu2+ peaks at 490 nm).
Figure 6. Photoluminescence spectra of the egg showing a strong blue luminescence with a maximum at 472 nm. This luminescence is attributed to the coating suspected to contain SrAl4O7:Eu2+ (Sr4Al14O25:Eu2+ peaks at 490 nm).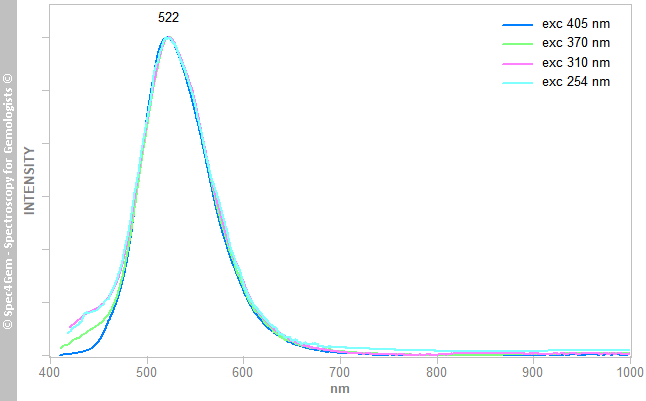 Figure 7. Photoluminescence spectra of the sphere showing a strong green luminescence with a maximum at 522 nm. This luminescence is attributed to the coating containing very likely SrAl2O4:Eu2+.
Figure 7. Photoluminescence spectra of the sphere showing a strong green luminescence with a maximum at 522 nm. This luminescence is attributed to the coating containing very likely SrAl2O4:Eu2+.Such luminescences and long lasting phosphorescences are well known and well studied[1],[2] since they are used by the industry to produce phosphorescent objects like the exit signs, watch hands, paints, etc. They rely on the luminescence of the REE (Rare Earth Element) while used to dop some materials. Common hosts for these REE, are the strontium aluminates that are produced with diferent forms such as SrAl2O4, SrAl4O7, SrAl12O19, Sr3Al2O6, Sr4Al14O25. The green luminescence at 522 nm is achieved by dopping SrAl2O4 with Eu2+ and it can be enhanced by co-dopping with Dy3+, generally in a lesser content than Eu2+. Regarding the blue luminescence at 472 nm, there are more options, either use another stontium aluminate such as SrAl4O7:Eu2+, Sr4Al14O25:Eu2+ forms peaking respectively at 470 and 490 nm or use the SrAl2O4:Eu2+ (as for the green) but co-dopped with La3+ or Tm3+ that respectively peaks at 470 and 485 nm.
Conclusion:
These two ornamental stones / objects are simply made of a dolomitic marble, then shaped and coated with a coating hosting strontium aluminate phosphors powders. There is a wide range of color available for these powders, search and browse on the internet for "strontium aluminate powder".
An experiment has been conducted to build some glow in the dark roads. The following photography is from the article: Singapore is testing glow-in-the-dark roads for better night visibility | Mashable.
[1] Effects of co-activator species and transition metal ions doping on structure and fluorescence properties of strontium aluminate
phosphors, Xiaowu Hu, Limin Wu, Hao Yin, Guizhi Li, Dongcai Guo, 2019, Journal of Materials Science: Materials in Electronics, 30, pp. 3804-3810, DOI: 10.1007/s10854-019-00664-y
[2] Eu2+ luminescence in strontium aluminates, D. Dutczak, T. Jüstel, C. Rondac and A. Meijerink, 2015, Physical Chemistry Chemical Physics, Royal Society of Chemistry, DOI: 10.1039/c5cp01095k


